 
The outer most shell is called valance shell and the electron in it are called valence electron. The charge on the ion tells you the number of. The number of electron in the outer most shell is not distributed 2(N) 2 formula. However, it is possible to remove electrons and not change the identity of an element. The number of electron in M shell 2N 2 = 2(3) 2 = 18 Etc, etc 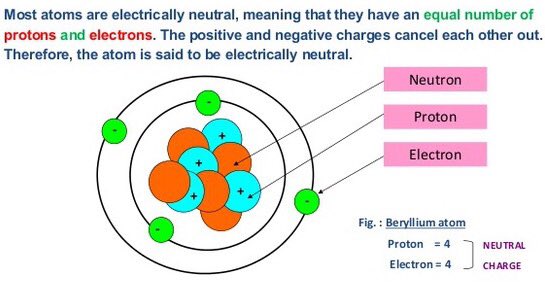
The number of electron in L shell 2N 2 = 2(2) 2 = 8 The number of electron in K shell 2N 2 = 2(1) 2 = 2 These positions are called energy levels or shell electrons are distributed among the shell according to 2(N) 2 formula. We know that electrons are revolving around the nucleus in different position. It is 1842 times heavier than electron.both the proton and neutron make the atomic mass of the atom. It mass is nearly equal to the mass of proton that is equal to 1.6x10 -27kg or 1.0086654 a.m.u. Hence the name Neutron is derived form the word neutral. Neutron is a neutral particle thus it has not any charge. The number of protons and electrons are equal in an atom. The amount of charge is 1.6x10 -19 coulomb. Both protons and neutrons have a mass of 1, while electrons have almost no mass. Proton is a particle which has positive charge.It is inside the nucleus. The negative charge of one electron balances the positive charge of one proton. Since atom has equal number of electrons and protons and they have equal and opposite charges hence they cancel their effect and atom becomes neutral. The amount of the charge is -1.6x10 -19 coulomb. Electron is a particle which has negative charge. It also a fundamental particle of the atom. This depends both on the fraction of charges and on the number and types of particles you have in one family of the standad model. A Short description about these particles is given below. There are theoretical reasons based on the standard model why charges of the electron (-e) and the quarks (e/3, 2e/3) fulfil certain relations. The nucleus consists of positively charged particles called protons and uncharged particles called neutrons. According to the classical Bohr model, atoms have a planetary type of structure that consists of a central nucleus surrounded by orbiting electrons. Iron, Al, cu, etc ATOMIC STRUCTUREĪn atom is the smallest particle of an element that retains the characteristics of that element. Between them neutron does not have any electric charge but. The smallest partical of the element, which remain the properties of the element.Ītom is a Greek word and its means is particle, so atom is smallest particle of the mater, which has properties of element. Electron, proton and neutron are the most important Subatomic particles which made atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
